August 10, 2018Press Release
Autoimmune Response Drives Vision Loss in Glaucoma
Media Contact
Mass Eye and Ear Communications
communications@meei.harvard.edu
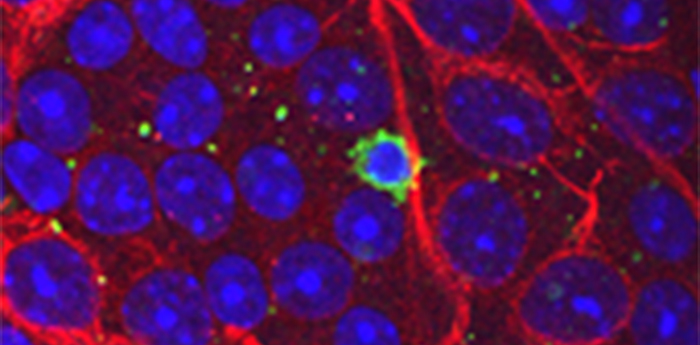 New research shows that immune cells developed from early exposure to bacteria contribute to vision loss in glaucoma patients
New research shows that immune cells developed from early exposure to bacteria contribute to vision loss in glaucoma patients
Boston, Mass. — A research team from Massachusetts Eye and Ear and MIT has shown that immune cells in the eye that developed in response to early exposure to bacteria are a key contributor to progressive vision loss from glaucoma, the second leading cause of irreversible blindness in the world. The findings, published online in Nature Communications, suggest that high pressure in the eye leads to vision loss by setting into motion an autoimmune response that attacks the neurons in the eye — similar to immune responses triggered by bacterial infections. The discovery of these immune cells also reveals a promising new target for future therapies to be developed for the blinding condition.
“Our work shows that there is hope for finding a cure for glaucoma, or even preventing its development entirely, if we can find a way to target this pathway,” said co-senior author Dong Feng Chen, M.D., Ph.D., a vision scientist at Mass. Eye and Ear and associate professor of ophthalmology at Harvard Medical School. “Current glaucoma therapies are designed solely to lower eye pressure; however, we’ve known that, even when patients with glaucoma are treated and their eye pressure returns to normal, they can still go on to have vision loss. Now, we know that stress from high eye pressure can initiate an immune response that triggers T cells to attack neurons in the eye.”
These findings shed light on a process that largely has remained a mystery ― until now. Glaucoma is a group of eye conditions that often culminate in irreparable damage to the optic nerve, which transmits visual information from the eye to the brain. Elevated eye pressure is known to be the most important risk factor for glaucoma, however, little was understood as to how and why patients with high eye pressure go on to develop permanent vision loss. Furthermore, some patients with glaucoma do not have elevated eye pressure, and some patients still experience optic nerve degeneration and subsequent vision loss even after their eye pressure is under control with treatment.
The authors of the Nature Communications study observed a new mechanism to explain the series of events leading to permanent vision loss from glaucoma. When pressure in the eye rises, it induces the expression of heat shock proteins, a family of proteins that develop in response to stressful conditions. This leads to a response from immune cells — memory T cells — that are programmed to respond to heat shock proteins. The memory T cells attack the neurons of the retina, leading to degeneration of the optic nerve and often permanent loss of vision. T cell responses are essential in the development of progressive vision loss following elevated eye pressure.
The research team observed an immune response to heat shock proteins both in mice and in human patients with glaucoma. The team first detected T cells in the retina of a mouse model of glaucoma, which motivated experiments to determine if the T cells played a role in neuron loss. The team studied three groups of mice with glaucoma — some without T cells, some without B cells, and some without T or B cells. Overwhelmingly, they observed a loss of neurons in the mice only if the mice contained functional T cells. More strikingly, development of glaucoma-inducing T cells required early exposure to bacteria; mice never exposed to bacteria (being raised in a “germ-free” facility) were free from glaucoma under elevated eye pressure.
The researchers also studied blood samples from patients with primary open angle glaucoma (POAG), the most common type of glaucoma. In humans, they observed T cells responses similar to the mice that were well over 5-fold higher in patients with POAG compared to samples from patients without POAG.
These findings open the door for the possibility of targeting T cells in the eye as a treatment to halt the progression of vision loss in glaucoma. Furthermore, heat shock proteins have been found in other conditions of the eye and the brain, which suggests that these findings could extend to other neurodegenerative conditions as well.
Pictured above: Elevated eye pressure caused T cell infiltration into the eye. Epifluorescence microscope of a flat-mounted retina of a glaucoma mouse showing an infiltrated T cell (green) into the mouse eye and resided next to retinal ganglion cells (red) – neurons which send long nerve fibers to form the optic nerve. Credit: Huihui Chen of Massachusetts Eye and Ear.
In addition to Dong Feng Chen, authors on the Nature Communications report include co-corresponding author Jianzhu Chen, Ph.D, of Massachusetts Institute of Technology, co-first authors Huihui Chen, of Second Xiangya Hospital and Massachusetts Eye and Ear/Harvard Medical School, Kin-Sang Cho of Massachusetts Eye and Ear/Harvard Medical School, and T.H. Khanh Vu, of Massachusetts Eye and Ear/Harvard Medical School and Leiden University Medical Center. Additional co-authors include Ching-Hung Shen, Mandeep Kaur, Mark T. Whary and James G. Fox of the Massachusetts Institute of Technology, Guochun Chen of Second Xiangya Hospital, Rose Mathew, Ahad Fazelat, Kameran Lashkari, Yingqian Li, Honghua Yo, Lanbo Yang, Joan Stein-Streilein of Massachusetts Eye and Ear/Harvard Medical School, M. Lisa McHam of Massachusetts Eye Health Service, Ngan Pan Bennett AU, Koyce Ka Yu TSE of City University of Hong Kong, Chi Him Eddie Ma of City University of Hong Kong and Boston Children’s Hospital/Harvard Medical School and Martine J. Jager of Leiden University Medical Center.
This research was supported by grants EY025913, EY027067, EY025259, NS038253 and AI69209 from the National Institutes of Health, 81200683 and 20120165 from the National Nature Science Foundation of China, P30-CA14051 from the National Cancer Institute and P30EY03790 from the National Eye Institute.
About Massachusetts Eye and Ear
Massachusetts Eye and Ear, founded in 1824, is an international center for treatment and research and a teaching hospital of Harvard Medical School. Specializing in ophthalmology (eye care) and otolaryngology-head and neck surgery (ear, nose and throat care), Mass. Eye and Ear clinicians provide care ranging from the routine to the very complex. Also home to the world's largest community of hearing and vision researchers, Mass. Eye and Ear has pioneered new treatments for blindness, deafness and diseases of the head and neck. Our scientists are driven by a mission to discover the basic biology underlying these conditions and to develop new treatments and cures. In the 2017-2018 "Best Hospitals Survey," U.S. News & World Report ranked Mass. Eye and Ear #1 in New England for eye (#4 in nation) and ear, nose and throat care (#2 in nation). For more information about life-changing care and research at Mass. Eye and Ear, please visit our blog, Focus, and follow us on Twitter and Facebook.
About Harvard Medical School Department of Ophthalmology
The Harvard Medical School Department of Ophthalmology (eye.hms.harvard.edu) is one of the leading and largest academic departments of ophthalmology in the nation. More than 400 full-time faculty and trainees work at nine Harvard Ophthalmology affiliate institutions, including Massachusetts Eye and Ear, Schepens Eye Research Institute of Massachusetts Eye and Ear, Massachusetts General Hospital, Brigham and Women’s Hospital, Boston Children’s Hospital, Beth Israel Deaconess Medical Center, Joslin Diabetes Center/Beetham Eye Institute, Veterans Affairs Boston Healthcare System, VA Maine Healthcare System, and Cambridge Health Alliance. Formally established in 1871, the department has been built upon a strong and rich foundation in medical education, research, and clinical care. Through the years, faculty and alumni have profoundly influenced ophthalmic science, medicine, and literature—helping to transform the field of ophthalmology from a branch of surgery into an independent medical specialty at the forefront of science.

